Top 10 Laboratory Chemicals Essential for Every Research Facility?
In the realm of scientific research, the importance of laboratory chemicals cannot be overstated. Dr. James Harmon, a seasoned expert in the field, once stated, "The right chemicals can make or break an experiment." This profound insight reflects the central role that laboratory chemicals play in achieving reliable results.
Every research facility relies on a wide variety of laboratory chemicals. From reagents used in tests to solvents that facilitate reactions, these substances are fundamental. The balance between quality and availability matters immensely. Inadequate supplies lead to delays and subpar results. Researchers often find themselves reflecting on the implications of their choices in chemical selection.
Chemicals used in laboratories should be chosen carefully. Quality impacts not just the experiments but the facility's reputation. Therefore, staying informed about the essential laboratory chemicals is crucial for every lab professional. Ultimately, the success of scientific endeavors hinges on the mindful approach to handling these pivotal substances.

Essential Solvents: The Backbone of Laboratory Processes
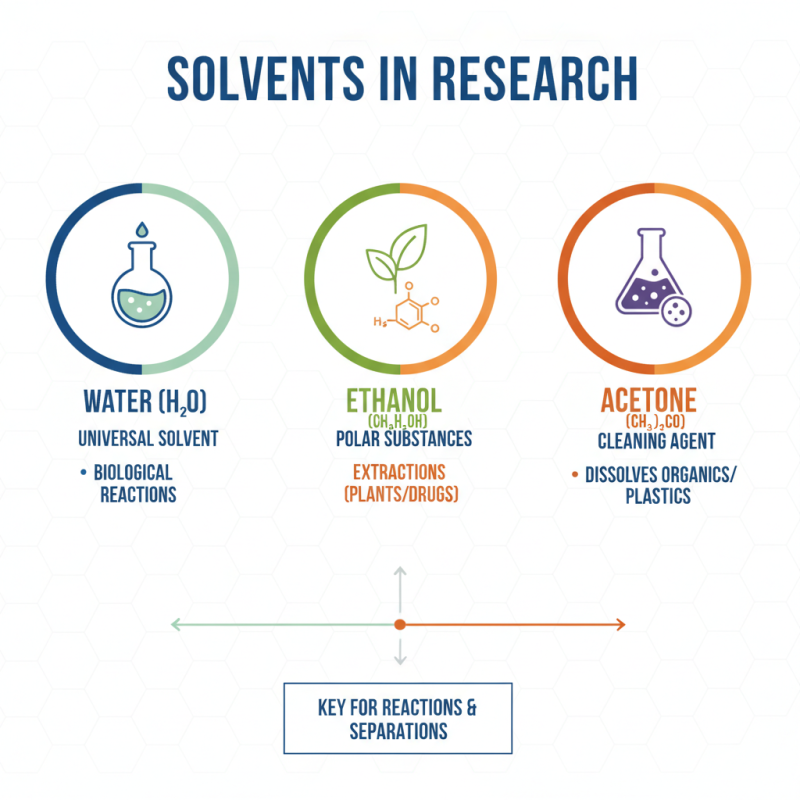
In any research facility, solvents play a pivotal role. They are used in various chemical reactions and extractions. Essential solvents like water, ethanol, and acetone are commonly used. Each solvent has unique properties that make it suitable for specific tasks. Water is universal, while ethanol is excellent for polar substances. Acetone effectively cleans and dissolves unwanted materials.
However, using solvents requires caution. Proper storage and disposal are crucial to maintain safety. Some solvents evaporate quickly, creating harmful vapors. Mixing different solvents can lead to unexpected reactions. Always label containers clearly. It is vital to understand each solvent's chemical properties and compatibility. Researchers sometimes overlook the importance of solvent purity. Impurities can jeopardize results, making meticulous preparation essential.
Solvents also have varying boiling points and polarities. This can impact experiments significantly. Researchers need to keep records of their solvent use. Tracking solvent quality over time is equally important. While solvents are indispensable, their management can be complex. Balancing efficiency and safety is an ongoing challenge.
Common Reagents: Key Players in Chemical Reactions
In any research facility, common reagents are vital. These chemicals act as the backbone of numerous experiments. A study published by the American Chemical Society noted that over 70% of chemical reactions rely on these essential compounds. The accuracy and success of laboratory work often hinge on these key players.
Common reagents like acids and bases intersect across various scientific fields. Acetic acid, for instance, is a versatile reagent in organic synthesis. Modifying its concentration can lead to different outcomes in reactions. On the other hand, sodium hydroxide serves as a strong base essential for titration processes. However, improper handling can lead to hazardous situations. Indeed, a report showed that lab accidents often stem from reagent misuse.
The importance of purity cannot be overstated. Impurities can skew results, leading to inaccurate conclusions. A study highlights that nearly 30% of all lab errors arise from contaminated reagents. This statistic underscores the need for rigorous quality checks and proper storage methods. Researchers must constantly reflect on their practices to maintain high standards and ensure reliable results.
pH Indicators: Crucial for Monitoring Acidity and Alkalinity
pH indicators play a vital role in laboratory settings. They help researchers monitor acidity and alkalinity in various solutions. This is crucial for experiments that require precise pH levels. According to a report by the American Chemical Society, pH levels can significantly affect the outcome of chemical reactions.
Many laboratories rely on liquid pH indicators. These solutions change color at specific pH levels. For example, phenolphthalein turns pink in basic solutions. However, the accuracy of pH indicators can vary. Environmental factors like temperature and light exposure can impact results. Therefore, lab personnel must consider these variables.
Solid indicators, like pH test strips, are also common. They provide a quick and portable method to check pH levels. A study from the Journal of Analytical Chemistry highlighted that these strips are less accurate than liquid indicators. They are, however, convenient for fieldwork. Despite their ease of use, they may lead to errors if not interpreted correctly. Researchers should always cross-check results with calibrated equipment.
Safety Chemicals: Ensuring a Safe Laboratory Environment

Creating a safe laboratory environment is crucial for research facilities. Safety chemicals play a vital role in minimizing hazards. According to industry reports, around 40% of laboratory accidents stem from improper handling of chemicals. This highlights the need for strict safety protocols.
A few essential safety chemicals include corrosive neutralizers, spill kits, and protective agents. Corrosive neutralizers help neutralize spills from acids and bases. Spill kits allow for quick cleanup of hazardous substances. Protective agents safeguard surfaces and equipment from damage. Having these on hand significantly reduces risks.
Tip: Always read and follow Material Safety Data Sheets (MSDS) for each chemical. Understanding the risks can prevent accidents.
Training staff is equally important. Employees should receive regular training on chemical handling and emergency protocols. Yet, some facilities overlook this crucial aspect. Reports show that labs without regular training are 30% more likely to experience incidents.
Tip: Keep training sessions engaging. Use real-life scenarios to enhance learning. This can reinforce safety behaviors among staff.
Storage and Handling: Best Practices for Laboratory Chemicals
Proper storage and handling of laboratory chemicals is crucial for safety and efficiency. Always label containers clearly. Use appropriate symbols for hazards and risks. Keep compatibilities in mind when arranging shelves. For instance, acids should be stored away from bases. It’s easy to overlook basics, like using secondary containment for liquids. This can be a game-changer in spill prevention.
Temperature control is another vital aspect. Some chemicals require refrigeration, while others need to be kept at room temperature. A well-organized storage area helps minimize accidents. Regularly review your inventory. Expired or unwanted chemicals should be disposed of properly. Ignoring this can lead to hazardous conditions. Adding shelves improves accessibility but can also create clutter if not managed well. Remember, the more organized your storage, the safer your lab becomes.
Top 10 Essential Laboratory Chemicals
Related Posts
-

How to Safely Handle Laboratory Chemicals in Your Home Lab
-

10 Essential Tips for Choosing Reliable Chemical Providers?
-
What is Chem Supply and Why is it Important for Industries?
-

Top 10 Essential Chemicals in Chemistry You Need to Know
-

Why Are Chemical Raw Materials Essential for Modern Manufacturing Processes?
-

How to Choose the Right Chemical Supply for Your Business Needs
Contact Us
We offer a just-in-time delivery solution supplying the highest quality materials to our customers. Get in touch today and learn how we can help your company.
Get in touch